# Lately in Science, not Fiction. Part 1
1. Researchers Made Artificial Cells That Can Replicate Themselves
“Scientists have a pretty good theory for how life on Earth began:
Meteorites that bombarded our planet brought simple carbon-based
compounds called amino acids (like discovery of air on comet 67P /
Churyumov-Gerasimenko and other elements = link*).
Eventually, slowly, these chemicals combined to make cells, which were then able to replicate and become the increasingly complex forms of life that we have today.
But researchers didn’t quite understand the mechanisms through which the earliest life forms evolved; though these cells were able to replicate, they were not yet alive.
Now a team of Japanese biologists has created artificial cells similar to those that might have first existed on Earth to better understand how they might have started to divide and evolve, according to a study published today in Nature Communications.
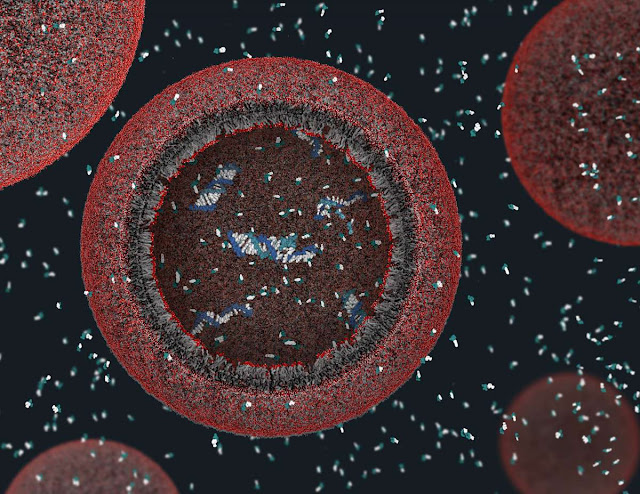
The researchers made a synthetic “protocell” made of DNA and proteins packaged inside lipids, which are fatty compounds meant to mimic the cell membrane. These spheres aren’t alive, but the DNA in them contains instructions to replicate under the right conditions.
By changing the pH of the spheres’ environment, the researchers were able to trigger the cells to divide. But the hard part was replenishing the spheres’ supplies so that they could start the division process over again, as real cells do. To work around this, the researchers designed the newly split synthetic cells to combine with other cell-like structures nearby. It worked — the spheres had three successful generations in the lab.”
Sources: nature.com and popsci.com
Eventually, slowly, these chemicals combined to make cells, which were then able to replicate and become the increasingly complex forms of life that we have today.
But researchers didn’t quite understand the mechanisms through which the earliest life forms evolved; though these cells were able to replicate, they were not yet alive.
Now a team of Japanese biologists has created artificial cells similar to those that might have first existed on Earth to better understand how they might have started to divide and evolve, according to a study published today in Nature Communications.
The researchers made a synthetic “protocell” made of DNA and proteins packaged inside lipids, which are fatty compounds meant to mimic the cell membrane. These spheres aren’t alive, but the DNA in them contains instructions to replicate under the right conditions.
By changing the pH of the spheres’ environment, the researchers were able to trigger the cells to divide. But the hard part was replenishing the spheres’ supplies so that they could start the division process over again, as real cells do. To work around this, the researchers designed the newly split synthetic cells to combine with other cell-like structures nearby. It worked — the spheres had three successful generations in the lab.”
Sources: nature.com and popsci.com
2. “Neural Net of Gaian Consciousness”~ Paul Stamets (mycologist)
+ story about his patent that could destroy MONSANTO
and change the World!
“The ecosystem is
tied together with an infused mass of mycellium, and they share
information and knowledge. Here’s - photos which shows the organization
of the computer internet... very mycellium-like in its form. There’s no
point-specific place where the internet can be totally disrupted.
The mycelial form also follows this archetype. I present to you the concept that the invention of the computer internet is just an extension of a previously proven biological model that has been refined over millions of years of experimentation.
It is no accident that we invented the computer internet at a time when we are in crisis, ecologically and politically, so we can share information and create countries without borders.”
The mycelial form also follows this archetype. I present to you the concept that the invention of the computer internet is just an extension of a previously proven biological model that has been refined over millions of years of experimentation.
It is no accident that we invented the computer internet at a time when we are in crisis, ecologically and politically, so we can share information and create countries without borders.”
* * *
More about Paul Stamets, whos name you should remember!
In 2006, a patent was granted to a man named Paul Stamets. Though Paul is the world’s leading mycologist, his patent has received very little attention and exposure.
The mycologist has figured out how to use mother nature’s own creations to keep insects from destroying crops. It’s what is being called SMART pesticides. These pesticides provide safe & nearly permanent solution for controlling over 200,000 species of insects - and all thanks to the ‘magic’ of mushrooms.
Monsanto’s chemical concoctions which are being sprayed all over farmers’ fields around the world are attributed to the large-scale bee die off. To say that new methods need to be implemented before it is too late is an understatement.
Sources:
Link to the patent we are speaking of: 7,122,176
All the patents Paul has applied for: Here
Paul Stamet’s Wikipedia page
More about Paul Stamets, whos name you should remember!
In 2006, a patent was granted to a man named Paul Stamets. Though Paul is the world’s leading mycologist, his patent has received very little attention and exposure.
The mycologist has figured out how to use mother nature’s own creations to keep insects from destroying crops. It’s what is being called SMART pesticides. These pesticides provide safe & nearly permanent solution for controlling over 200,000 species of insects - and all thanks to the ‘magic’ of mushrooms.
Monsanto’s chemical concoctions which are being sprayed all over farmers’ fields around the world are attributed to the large-scale bee die off. To say that new methods need to be implemented before it is too late is an understatement.
Monsanto presently generates $16 billion dollars per year (as reported in 2014), therefore you can be certain they do not want anything interrupting that flow of revenue. Such income gives them nearly limitless resources and abilities to suppress information that may be damaging their reputation."
Sources:
Link to the patent we are speaking of: 7,122,176
All the patents Paul has applied for: Here
Paul Stamet’s Wikipedia page

3. Oldest tree in Scotland is 'CHANGING SEX' after 5,000 years!
"The oldest tree in Scotland has started to change sex, it has been claimed.
The 5,000 year old Fortingall Yew, in Perthshire, Scotland, has for hundreds of years been recorded as 'male', meaning it produces pollen, whereas female yews produce red berries.
But in a 'rare and unusual phenomenon' baffling scientists, it has started to sprout red seeds, suggesting that part of the tree is changing gender.
Max Coleman of Royal Botanic Garden Edinburgh, who spotted the 'three red berries', said that the phenomenon could be caused by 'environmental stress'.
He said: 'It's a rare occurrence... rare and unusual and not fully understood.
'It's thought that there's a shift in the balance of hormone-like compounds that will cause this sex-change.
'One of the things that might be triggering it is environmental stress.'
The tree is otherwise healthy, but Mr Coleman said it would be monitored closely for further changes."
The gnarled tree is in an old church yard, and is difficult to age because its heartwood - the wood in the centre of the tree - has long rotted away.
Scientists have been able to estimate the tree's age by comparing its current size with measurements taken in the 1700s.
Mr Coleman wrote in his blog: 'Yews are normally either male or female and in autumn and winter sexing yews is generally easy.
'Males
have small spherical structures that release clouds of pollen when they
mature. Females hold bright red berries from autumn into winter.
'It
was, therefore, quite a surprise to me to find a group of three ripe
red berries on the Fortingal yew this October when the rest of the tree
was clearly male.
'Odd as it may seem, yews, and many other conifers that have seperate sexes, have been observed to switch sex.
'Normally
this switch occurs on part of the crown rather than the entire tree
changing sex. In the Fortingall Yew it seems that one small branch in
the outer part of the crown has switched and now behaves as female.'
He
said the three seeds had been collected and would be included in a
major project to 'conserve the genetic diversity of yew trees' by
planting them out at the Botanic Garden."
Source: dailymail.co.uk
4. DARPA: We Are Engineering the Organisms That Will Terraform Mars!
"It’s no secret that the Defense Advanced Research Projects Agency is investing heavily in genetic engineering and synthetic biology. Whether that excites or terrifies you depends on how you feel about the military engineering totally new life forms. If you’re in the excitement camp, however, here’s a nugget for you: DARPA believes that it's on the way to creating organisms capable of terraforming Mars into a planet that looks more like Earth.
The goal of terraforming Mars would be to warm up and potentially thicken its atmosphere by growing green, photosynthesizing plants, bacteria, and algae on the barren Martian surface. It’s a goal that even perpetual techno-optimists like Elon Musk think isn’t going to happen anytime soon, but it’s a goal that DARPA apparently already has its eyes on.
“For the first time, we have the technological toolkit to transform not just hostile places here on Earth, but to go into space not just to visit, but to stay,” Alicia Jackson, deputy director of DARPA’s new Biological Technologies Office said Monday at a DARPA-hosted biotech conference. As she said this, Jackson was pointing at an artist's rendering of a terraformed Mars.
So what’s this technological toolkit she’s talking about? For the last year, Jackson’s lab has been working on learning how to more easily genetically engineer organisms of all types, not just e. coli and yeast, which are most commonly used in synthetic biology projects.
“There are anywhere from 30 million to 30 billion organisms on this Earth. We use two right now for engineering biology,” she said. “I want to use any organism that has properties I want—I want to quickly map it and quickly engineer it. If you look at genome annotation software today, it’s not built to quickly find engineer able systems [and genes]. It’s built to look for an esoteric and interesting thing I can publish an academic paper on.”
DARPA and some of its research partners have created software called DTA GView, which Jackson calls the “Google Maps of genomes.” At the conference, she pulled up the genomes of several organisms on the program, which immediately showed a list of known genes and where they were located in the genome.
“This torrent of genomic data we’re now collecting is awesome, except they sit in databases, where they remain data, not knowledge. Very little genetic information we have is actionable,” she said. “With this, the goal is to, within a day, sequence and find where I can best engineer an organism.”
The goal is to essentially pick and choose the best genes from whatever form of life we want and to edit them into other forms of life to create something entirely new. This will probably first happen in bacteria and other microorganisms, but it sounds as though the goal may to do this with more complex, multicellular organisms in the future.
The utility of having such a capability is pretty astounding: Jackson threw out goals of eradicating vector-borne illnesses, which obviously sounds lovely and utopian. But perhaps more interesting is DARPA’s plan to use specifically engineered organisms to help repair environmental damage. Jackson said that after a natural or man-made disaster, it’d be possible to engineer new types of extremophile organisms capable of surviving in a scarred wasteland. As those organisms photosynthesized and thrived, it would naturally bring that environment back to health, she said.
And that’s where terraforming Mars comes in. With enough practice turning Earth’s damaged landscapes back into places hospitable for life, Jackson thinks we’ll have what it takes to eventually try to colonize the solar system. This is something that obviously doesn’t even really have a timeline, it’s technology that’s in its infancy, and much of the work being done is classified—but the implications are exciting nonetheless.
“After a manmade or natural disaster, we can think about recovering the environment,” she said. “These are the tools that, for the first time, are allowing us to go after that problem.”
Source: motherboard.vice.com

5. Reprogrammed stem cells work as well as those from embryos
"Researchers who hope to use stem cells—the unspecialized cells that produce all of our tissues—to treat diseases face a dilemma. Stem cells from embryos (ES cells) could provide a wealth of new cells but spark ethical objections. Stem cells produced from adult cells (so-called induced pluripotent stem [iPS] cells) avoid the ethical difficulties, but some scientists have questioned whether they are as powerful as ES cells. A new study suggests that the two types of stem cells are equivalent and may help soothe worries about the capabilities of iPS cells.
“I think it was a very well done study, and it will ease some of the concerns about ES cells versus iPS cells,” says stem cell biologist Joseph Wu of the Stanford University School of Medicine in Palo Alto, California, who wasn’t connected to the study. Stem cell biologist William Lowry of the University of California, Los Angeles, agrees. “It’s probably going to be a lot easier to go forward with iPS cells because of studies like this.”
ES cells can specialize into any type of cell in the body, but they are controversial because obtaining them destroys embryos. In 2006, researchers found a possible workaround. Adding four genes to a specialized adult cell—such as a skin cell from a patient—can convert it into an iPS cell. These cells seem to have similar abilities as ES cells in the lab, and iPS cells don’t rouse the ethical objections of ES cells. Such reprogrammed cells are now widely used by stem cell labs around the world, and the scientist who figured out how to make them, Shinya Yamanaka of Kyoto University in Japan, won the Nobel Prize in Physiology or Medicine in 2012 for the discovery.
Problem solved, right? Not quite. Some researchers have questioned
whether iPS cells measure up to ES cells. Different genes are switched
on in iPS cells, some studies have found, raising the possibility that
the two types of stem cells aren’t equally capable. “There is still
controversy out there about how similar they are,” says stem cell
researcher Konrad Hochedlinger of Massachusetts General Hospital in
Boston. Determining whether ES and iPS cells are comparable is important
because clinical trials of both kinds of cells are underway.
Hochedlinger and his colleagues note that several factors could explain why iPS cells and ES cells show different patterns of gene activity. One is the source of the cells. The embryo used to derive an ES cell is not genetically identical to the donor of the cell that is transformed into an iPS cell, so researchers expect some discrepancies in gene activity. The sex of the stem cells could also be significant, because activity of some genes varies between males and females.
To try to rule out these factors, the scientists obtained two lines of ES cells that were male. They allowed cells from each line to mature and then coaxed these cells to transform into iPS cells. Thus, the iPS cells genetically matched their ES cell parents. When the researchers measured gene activity, they found that the iPS cells were more similar to the ES cells they descended from than to each other, suggesting that the source of the cells explains many of the disparities researchers had noted between the two stem cell types.
The iPS cells the researchers created weren’t identical to their ES cell parents, however. Hochedlinger and colleagues pinpointed 49 genes whose activity consistently differed between iPS and ES cells. To gauge whether these differences altered the cells’ capabilities, the scientists analyzed two of the genes. One helps cells imbibe glucose, whereas the other helps cells break it down. Even though both genes were less active in iPS cells, the cells were just as efficient as ES cells at absorbing and digesting glucose. At least when it comes to these two genes, the researchers conclude, the two types of cells are functionally equivalent.
The ultimate test of a stem cell is its ability to produce different cell types. ES cells and iPS cells were equally good at specializing into a variety of nervous system cells, the researchers report online today in Nature Biotechnology. They also ran a standard test that measures the cells’ ability to produce the three major cell lineages in the body and found no differences. “The two cell types appear functionally indistinguishable based on the assays we used,” Hochedlinger says.
The paper won’t silence all doubts about iPS cells, says stem cell biologist Andras Nagy of the Lunenfeld-Tanenbaum Research Institute of Mount Sinai Hospital in Toronto, Canada. The fact that the researchers made their iPS cells from cells that matured in the lab from ES cells, instead of from the cells of a patient or donor, means that they still might carry traces of the ES cells’ gene activity patterns. “It’s a nice paper, but still there is a little bit of reservation about how broad the conclusion can be,” he says.
Source: sciencemag.org
6. Gene Editing Is Now Cheap and Easy — Does World Is Prepared for it?
Discovered by scientists only a few years ago, CRISPRs are elements of an ancient system that protects bacteria and other single-celled organisms from viruses, acquiring immunity to them by incorporating genetic elements from the virus invaders. CRISPRs evolved over millions of years to trim pieces of genetic information from one genome and insert it into another. And this bacterial antiviral defense serves as an astonishingly cheap, simple, elegant way to quickly edit the DNA of any organism in the lab.
Until recently, editing DNA required sophisticated labs, years of experience, and many thousands of dollars. The use of CRISPRs has changed all that. CRISPRs work by using an enzyme — Cas9 — that homes in on a specific location in a strand of DNA. The process then edits the DNA to either remove unwanted sequences or insert payload sequences. CRISPRs use an RNA molecule as a guide to the DNA target. To set up a CRISPR editing capability, a lab only needs to order an RNA fragment (costing about $10) and purchase off-the-shelf chemicals and enzymes for $30 or less.
Because CRISPR is cheap and easy to use, it has both revolutionized and democratized genetic research. Hundreds, if not thousands, of labs are now experimenting with CRISPR-based editing projects. A race is on between the major research institutions to file CRISPR-technique patents. Research dollars, both public and private, are pouring into CRISPR projects. Meanwhile, a panoply of leading geneticists — including one of the developers of the CRISPR technology — has urged for a moratorium on alterations to the human germ line until the implications of messing with human DNA are further studied and safeguards put in place.
Changing human DNA creates, for scientists and humanity, a frightening ethical grey zone. On the one hand, for the many millions of poor souls suffering from diseases arising from genetic defects, CRISPR and the research it fuels could mean finding a cure for their problem in their lifetimes. On the other hand, changing the human germ line is incredibly risky without much better knowledge of how our DNA actually works.
Though scientists now commonly sequence human DNA, they still struggle to understand how the different pieces of the human genome work together. For example, until recently, scientists thought that much of our genetic material was useless and served no purpose. They called it “junk” DNA. In a previous era, they might have considered editing the junk out of our genes.
Now, research is emerging showing that junk DNA plays a key role in regulating genetic expression (effectively turning various genes on and off), regulation that is fundamental to the biological processes that govern our bodies and our endocrine systems. What if a well-intentioned researcher develops a cure for one of these diseases and shares it with thousands of sufferers before realizing that the cure is far worse than the disease and that the side effects are painful — or even deadly — and easily spread from person to person?
Such a scenario could arise through good intent. But in the hands of evil biohackers, these powerful and simple tools are a cause for alarm. A smart biohacker could alter the influenza genome, for example, to make it more potent, setting off an epidemic that kills hundreds of millions of people. Though a nuclear weapon can cause tremendous long-lasting damage, the ultimate biological doomsday machine is bacteria, because they can spread so quickly and quietly.
No one is prepared for an era when editing DNA is as easy as editing a Microsoft Word document. The government does not have any regulations on editing human DNA. The ethical concerns have not been fleshed out. There is no centralized risk-management inventory, listing which labs are doing what with CRISPR. It’s all rather terrifying.
Rarely do I argue that a moratorium on technological progress is the prudent course. But the stakes in the case of CRISPR are so high that I believe a blanket moratorium is the only course. Yes, rogue scientists may nonetheless continue working at modifications on the human germ line; and that could endow them with a first-mover advantage and unfair knowledge. But such a moratorium could be as effective as the global moratorium on the cloning of humans has been: at the least, scientists such as those who engineered the human embryos in China would become international pariahs rather than being celebrated for publishing papers in prestigious publications."
Source: singularityhub.com
Important to remember:
1. Genetics (work of Nature or humans) = source of all myths or/and conspiracy theories!
2. Not only humans fucked up with genes. Mother Nature too!
Hochedlinger and his colleagues note that several factors could explain why iPS cells and ES cells show different patterns of gene activity. One is the source of the cells. The embryo used to derive an ES cell is not genetically identical to the donor of the cell that is transformed into an iPS cell, so researchers expect some discrepancies in gene activity. The sex of the stem cells could also be significant, because activity of some genes varies between males and females.
To try to rule out these factors, the scientists obtained two lines of ES cells that were male. They allowed cells from each line to mature and then coaxed these cells to transform into iPS cells. Thus, the iPS cells genetically matched their ES cell parents. When the researchers measured gene activity, they found that the iPS cells were more similar to the ES cells they descended from than to each other, suggesting that the source of the cells explains many of the disparities researchers had noted between the two stem cell types.
The iPS cells the researchers created weren’t identical to their ES cell parents, however. Hochedlinger and colleagues pinpointed 49 genes whose activity consistently differed between iPS and ES cells. To gauge whether these differences altered the cells’ capabilities, the scientists analyzed two of the genes. One helps cells imbibe glucose, whereas the other helps cells break it down. Even though both genes were less active in iPS cells, the cells were just as efficient as ES cells at absorbing and digesting glucose. At least when it comes to these two genes, the researchers conclude, the two types of cells are functionally equivalent.
The ultimate test of a stem cell is its ability to produce different cell types. ES cells and iPS cells were equally good at specializing into a variety of nervous system cells, the researchers report online today in Nature Biotechnology. They also ran a standard test that measures the cells’ ability to produce the three major cell lineages in the body and found no differences. “The two cell types appear functionally indistinguishable based on the assays we used,” Hochedlinger says.
The paper won’t silence all doubts about iPS cells, says stem cell biologist Andras Nagy of the Lunenfeld-Tanenbaum Research Institute of Mount Sinai Hospital in Toronto, Canada. The fact that the researchers made their iPS cells from cells that matured in the lab from ES cells, instead of from the cells of a patient or donor, means that they still might carry traces of the ES cells’ gene activity patterns. “It’s a nice paper, but still there is a little bit of reservation about how broad the conclusion can be,” he says.
Source: sciencemag.org
6. Gene Editing Is Now Cheap and Easy — Does World Is Prepared for it?
"In April 2015, a paper by Chinese scientists about their attempts to
edit the DNA of a human embryo rocked the scientific world and set off a
furious debate. Leading scientists warned that altering the human germ
line without studying the consequences could have horrific consequences.
Geneticists with good intentions could mistakenly engineer changes in
DNA that generate dangerous mutations and cause painful deaths.
Scientists — and countries — with less noble intentions could again try
to build a race of superhumans.
Human DNA is, however, merely one of many commercial targets of ethical concern. The DNA of every single organism — every plant, every animal, every bacterium — is now fair game for genetic manipulation. We are entering an age of backyard synthetic biology that should worry everybody. And it is coming about because of CRISPRs: clustered regularly interspaced short palindromic repeats.
Human DNA is, however, merely one of many commercial targets of ethical concern. The DNA of every single organism — every plant, every animal, every bacterium — is now fair game for genetic manipulation. We are entering an age of backyard synthetic biology that should worry everybody. And it is coming about because of CRISPRs: clustered regularly interspaced short palindromic repeats.
Discovered by scientists only a few years ago, CRISPRs are elements of an ancient system that protects bacteria and other single-celled organisms from viruses, acquiring immunity to them by incorporating genetic elements from the virus invaders. CRISPRs evolved over millions of years to trim pieces of genetic information from one genome and insert it into another. And this bacterial antiviral defense serves as an astonishingly cheap, simple, elegant way to quickly edit the DNA of any organism in the lab.
Until recently, editing DNA required sophisticated labs, years of experience, and many thousands of dollars. The use of CRISPRs has changed all that. CRISPRs work by using an enzyme — Cas9 — that homes in on a specific location in a strand of DNA. The process then edits the DNA to either remove unwanted sequences or insert payload sequences. CRISPRs use an RNA molecule as a guide to the DNA target. To set up a CRISPR editing capability, a lab only needs to order an RNA fragment (costing about $10) and purchase off-the-shelf chemicals and enzymes for $30 or less.
Because CRISPR is cheap and easy to use, it has both revolutionized and democratized genetic research. Hundreds, if not thousands, of labs are now experimenting with CRISPR-based editing projects. A race is on between the major research institutions to file CRISPR-technique patents. Research dollars, both public and private, are pouring into CRISPR projects. Meanwhile, a panoply of leading geneticists — including one of the developers of the CRISPR technology — has urged for a moratorium on alterations to the human germ line until the implications of messing with human DNA are further studied and safeguards put in place.
Changing human DNA creates, for scientists and humanity, a frightening ethical grey zone. On the one hand, for the many millions of poor souls suffering from diseases arising from genetic defects, CRISPR and the research it fuels could mean finding a cure for their problem in their lifetimes. On the other hand, changing the human germ line is incredibly risky without much better knowledge of how our DNA actually works.
Though scientists now commonly sequence human DNA, they still struggle to understand how the different pieces of the human genome work together. For example, until recently, scientists thought that much of our genetic material was useless and served no purpose. They called it “junk” DNA. In a previous era, they might have considered editing the junk out of our genes.
Now, research is emerging showing that junk DNA plays a key role in regulating genetic expression (effectively turning various genes on and off), regulation that is fundamental to the biological processes that govern our bodies and our endocrine systems. What if a well-intentioned researcher develops a cure for one of these diseases and shares it with thousands of sufferers before realizing that the cure is far worse than the disease and that the side effects are painful — or even deadly — and easily spread from person to person?
Such a scenario could arise through good intent. But in the hands of evil biohackers, these powerful and simple tools are a cause for alarm. A smart biohacker could alter the influenza genome, for example, to make it more potent, setting off an epidemic that kills hundreds of millions of people. Though a nuclear weapon can cause tremendous long-lasting damage, the ultimate biological doomsday machine is bacteria, because they can spread so quickly and quietly.
No one is prepared for an era when editing DNA is as easy as editing a Microsoft Word document. The government does not have any regulations on editing human DNA. The ethical concerns have not been fleshed out. There is no centralized risk-management inventory, listing which labs are doing what with CRISPR. It’s all rather terrifying.
Rarely do I argue that a moratorium on technological progress is the prudent course. But the stakes in the case of CRISPR are so high that I believe a blanket moratorium is the only course. Yes, rogue scientists may nonetheless continue working at modifications on the human germ line; and that could endow them with a first-mover advantage and unfair knowledge. But such a moratorium could be as effective as the global moratorium on the cloning of humans has been: at the least, scientists such as those who engineered the human embryos in China would become international pariahs rather than being celebrated for publishing papers in prestigious publications."
Source: singularityhub.com
Important to remember:
1. Genetics (work of Nature or humans) = source of all myths or/and conspiracy theories!
2. Not only humans fucked up with genes. Mother Nature too!
 |
| "Werewolf syndrome" |
Zhang Hongming, from Chongqing (south-west China), suffers from rare congenital giant pigmented nevus, commonly know as giant furred moles.
Zhang was born with black birthmarks on his body but due to family poverty he didn’t receive any treatment. Over the years he has faced much abuse and mocking and been nicknamed things such as ‘monkey’, ‘beast’ and 'chimp’.
Fortunately ,Hua Mei Plastic Surgery Hospital' agreed to provide free treatment to Zhang after hearing his story. He has now undergone the first of six procedures to remove the hair and moles.
 |
| Black Albino (unusual genetic anomalies): |
 |
| E.T. from India (link*) |
 |
Genetic Disorder: “An Illness Caused By Abnormalities In The Genome”(+ Link) |
 |
"Hypohidrotic Ectodermal Dysplasia"
|
 |
"Lamellar Ichthyosis" (reptile's syndrome) |








Komentarze
Prześlij komentarz